Drugs comprise the major treatment modality of scientific medicine. According to data from the CDC, nearly half of Americans take at least one prescription drug regularly for chronic health problems. (1) Sadly, many people don’t realize that the drugs they’re taking could be making their condition worse.
Most drugs don’t cure illness. They just suppress symptoms. Unfortunately, drugs also suppress functions.
The Problem with Prescription Drugs
For example, many people take ibuprofen or other non-steroidal anti-inflammatory drugs (NSAIDs) to cope with arthritis and inflammatory conditions. While NSAIDs are effective in reducing pain and inflammation in the short-term, they are also known to reduce blood flow to cartilage. Since blood carries all of the nutrients and immune substance necessary for tissue repair, NSAIDs can actually worsen the original problem when taken chronically.
The second problem is that, by definition, drugs correct a specific imbalance by causing at least one other and often several other imbalances. When a drug is introduced into the body to address a malfunction in one biochemical pathway, that drug inevitably interacts with many other pathways.
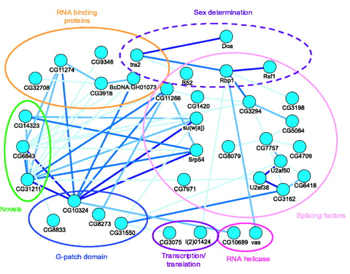
The mapping of these pathways in recent genetic research underscores the danger of pharmaceutical drugs. The diagram below shows the interactions among a small set of cellular proteins found in a fruit fly. Proteins encased in ovals are grouped according to specific pathway functions. Connecting lines indicate protein-protein interactions. Protein interconnections among the different pathways reveal how interfering with one protein may produce profound “side effects” upon other related pathways. (2)
Complicating the phenomenon of so-called “side effects” is that biological systems are redundant. The same protein molecule may be used in several different systems of the body, but it has a completely different function in each of them.
Histamine is a perfect example of this. Histamine is a chemical that initiates the cell’s stress response. When histamine is present in the bloodstream of the arms and legs, it starts a local inflammatory reaction in those tissues. But if histamine is present in the blood vessels of the brain, it enhances the growth and function of specialized neurons there.
One of the most amazing features of the body’s signaling system is its specificity. When you have a poison oak rash on your arm, histamine is released in that specific area only to activate an inflammatory response to the allergen. Likewise, if you’re under significant stress, histamine is released only in the brain to enhance the function of neurons.
It affects histamine receptors wherever they are located throughout the whole body. So, while the antihistamine will curb the blood vessels’ inflammatory response and reduce the allergic symptoms of the rash, it will also enter the brain and affect nerve function—which causes drowsiness.
The recent hormone replacement therapy (HRT) debacle is a tragic example of the inherent risks of pharmaceutical drugs. Estrogen is best known for its function on the female reproductive system. However, more recent studies have shown that estrogen also plays an important role in the normal function of blood vessels, the heart and the brain. That’s why synthetic estrogen hormones that were prescribed to alleviate menopausal symptoms ended up causing cardiovascular disease and neural dysfunctions such as strokes.
Another Problem with Prescription Drugs: Money and Conflicts of Interest
Massive conflicts of interest frequently exist between researchers, doctors, and the pharmaceutical industry in the U.S. and abroad. Studies funded by pharmaceutical companies are more likely to show positive results for the drug, and the veracity of clinical trials (which are the basis of approval of new drugs by the FDA) has been questioned because of three major flaws: (3)
- Conflicts of interest on the part of researchers and investigators
- Inappropriate involvement of research sponsors (drug companies) in study design and management
- Publication bias in disseminating results (if a study has negative results, the drug company doesn’t publish it)
When researchers fail to disclose their financial conflicts of interest, it can lead to catastrophic results. For example, consider Dr. Joseph Biederman, a world-renowned child psychiatrist at Harvard. Dr. Biederman accepted at least $1.6 million in consulting fees from drug makers from 2000 to 2007, but did not disclose any of this income to Harvard University officials. Dr. Biederman’s work directly contributed to a dramatic increase in the diagnosis of pediatric bipolar disorder and a concurrent rise in the use of powerful antipsychotic medicines in children. This is highly problematic when you consider that children are susceptible to weight gain and metabolic problems caused by the antipsychotic drugs used to treat pediatric bipolar disorder, and when you consider evidence suggesting that these drugs may cause permanent changes to the structure and function of the brain. (4, 5)
The research of Dr. Biederman’s group, which has served as the basis for the rise in bipolar diagnoses and antipsychotic use in children, has been widely criticized by other psychiatrists and researchers. More broadly, psychiatrists have said that revelations of undisclosed payments from drug makers to leading researchers are especially damaging for psychiatry.
“The price we pay for these kinds of revelations is credibility, and we just can’t afford to lose any more of that in this field,” said Dr. E. Fuller Torrey, executive director of the Stanley Medical Research Institute, which finances psychiatric studies. “In the area of child psychiatry in particular, we know much less than we should, and we desperately need research that is not influenced by industry money.”
I couldn’t have said it better myself.
The Number of Americans Who Take Prescription Drugs
As I mentioned above, nearly half of Americans take prescription drugs regularly. (1) In fact, according to the CDC:
- More than 48 percent of those surveyed used at least one prescription drug within the last 30 days
- Twenty-four percent used three or more
- Nearly 13 percent used a whopping five or more
Drug therapy is involved in nearly 74 percent of physician office visits and in more than 80 percent of visits to hospital emergency departments.
Many of the most commonly prescribed drugs (like statins and proton pump inhibitors or PPIs, for example) reveal a weakness of the mainstream medical model: its tendency to treat the symptom or effect of an illness rather than the cause. Unfortunately for patients, doing this can actually make things worse, not better. Functional Medicine, by contrast, addresses the root cause of an illness instead of simply suppressing symptoms with drugs. You can find out more about this medical paradigm in my article “What Is Functional Medicine?”
Major changes are definitely needed in the way we manage health in this country, but luckily, we don’t have to wait around for that to happen. As individuals, we can take responsibility for our own health using diet, exercise, and lifestyle changes, and we can take action to reduce stress and promote emotional and psychological well-being. And, when we’re struggling to make those changes or address underlying health conditions, we can seek the support of a trained health coach or Functional Medicine practitioner.
Better supplementation. Fewer supplements.
Close the nutrient gap to feel and perform your best.
A daily stack of supplements designed to meet your most critical needs.



Thank all of you so much for these comments. I am a former US Navy Corpsman (medic) who spent three of five years with the US Marines at Camp Lejeune, MCB in NC. We had a small satellite pharmacy and could not prescribe drugs for every whim. Captain Wallin, MSC, USN, and Commander Babb, MSC, USN, made it our priority to teach preventive measures instead of handing out drugs. Captain Wallin would have done so even with a massive budget.
There were so many times while I was in the midst of teaching a Marine how to eliminate the underlying cause of his/her condition, that the Marine would simply say to me, “Can’t you just give me a pill, Doc?” Those words were the bane of my existence! I would often respond with, “I’m trying to teach you how to overcome your illness and not take drugs!” In the last twelve years since my honorable discharge, I have taken one dose of medication–two sudafed tablets! I’ve not taken any over-the-counter nor prescription drugs, other than the one dose. and I’m ashamed for even taking that one!
In my last physical exam, a pre-op once-over, my doctor told me that I was in great health, no blood in my stool, no protein in my urine, my blood pressure was 95 / 65, and that he expected me to heal rapidly after my surgery (meniscus tear). He then told me to be sure to schedule an appointment in six months and he’d get me started on “preventive medication” for my colon and heart. I asked him why I would need those medications, and he replied, “Because the body changes and so do its needs.” If my only weakness had been Kryptonite (which would have obviated my surgery), the man would have been a smoking, smug cinder sitting in a smoldering leather chair! I asked the doctor why, if I was in great health, would I need to start taking preventive measures in as little as six months, only to offset my systemic balance by adding toxic chemicals which would store in my lipids, nerves, intestines and colon. The doctor was speechless for about six seconds, and at one point he opened his mouth to speak and then closed it and continued looking at me. Finally he spoke and said “If you experience any significant changes in your health, please schedule an appointment.”
The lesson here is that if a patient takes some time to educate her/himself about health and nutrition, and s/he makes the doctor aware of it, the doctor is less likely to slam the patient with a drug regimen. (I also made the doctor watch as I dropped his prescription for whateveritwas in the trash can.) Doctors want to go on being doctors; there are more of them than are actually needed, like lawyers. Doctors know what it takes to be truly healthy, and they also know that many of their patients lack the motivation to bothered with things like exercise, proper diet and stress management. Money is also a factor of course. If money were no object, I’d consume nothing but organic everything, drink oxygenated, purified water, sleep in a hyperbaric chamber, like the late Michael Jackson, and live above the pollution, like Tom, Kati and Suri Cruise. But money is an object, a vey dear one, and until such time that I’m making twenty million per picture (I’m an actor), I will continue to avoid all OTCs and prescription drugs, food coloring and preservatives, and will continue to filter my tap water with chlorine- and iodine- removing filters.
One of the best summaries I’ve ever read for the notion of taking pharmaceutical drugs or OTCs as a life-choice comes from Kevin Trudeau–“Were you born with an asprin deficiency?” LOL Words to live by!
Hello,
I’m writing / compiling a booklet describing the FDA-reported adverse events for the most popular prescription drugs – written in plain English. I feel that if people are presented with the real side effects and adverse events of these drugs in a way that is easily understood they would be less likely to endanger their own lives and the lives of those they love by taking drugs just because they have seen them on TV or been prescribed them by their doctor.
The booklet I’m working on is a compilation and organization of the top reported adverse events reported to the FDA in connection to the top 100 most prescribed and highest grossing prescription drugs.
In order to help market this book once completed I am trying to get as many people as possible to fill out a short survey. I’ve put it up on my website at PrescriptionDrugProblems.com. Those peple who fill out the survey will receive a free pre-release copy of this E-book as soon as it’s done.
I was wondering if you could mention this on your blog and do the survey yourself.
Thank you for your time.
Best,
Meridith
An interesting side note on histamine: Did you know that there is scientific evidence of histamine as an anti-cancer agent? I worked for a company who tried to market histamine to treat cancer. Isn’t it scary then that so many people are chronically on anti-histamines? Big problem for pharmaceutical companies is the issue of patents. Natural occuring chemicals such as histamine cannot be patented, therefore, ultimately there is no money in it. They will not try to develop a drug which may cure cancer, but have no patent protection. When trying to decide between which compound to develop into drugs, the factors used in the decision making process only uses $$$ made, not lives saved.
Yes, it is scary. Unfortunately there are so many examples of exactly the phenomenon you describe. Another one that I’m writing a series of articles about right now (first one is here) is the rampant use of acid-suppressing drugs for heartburn and GERD. Stomach acid is vital to our health and low stomach acid is associated with impaired nutrient absorption and increased risk of gastric cancer. Yet those drugs are now available over-the-counter and people are popping them like candy.
A very interesting post. In Poland, for instance, the country I am from, doctors will put you on antibiotics at the signs of a mild cough and a temperature. Just in case. To be on the safe side. You don’t even need to ask fot it. On the contrary, more often you have ask not to be given antibiotics but then there isn’t much of an alternative you can expect from your doctor instead.
And as far as I know, this is a standard protocol throughout the entire EU. I sometimes think that contemporary medicine has ceased to cure the coause of your disease but turned into a drug-selling industry with doctors acting like salespeople. In addition to that most of TV commercials are for non-prescription drugs. These are no longer recommended as remedies to health problems but as substances that will improve the quality of your everyday life. In other words, you should take a pill to avoid getting sick. Avarage Europeans are as helpless as they can get against all this misinformation.
Hey Chris,
For the most part, I agree that meds are overprescribed across the board. I think it’s a big mistake to blame big pharma exclusively though, since really, consumers speak with their money, and basically scream: Give me a pill, I’m too tired, too busy, too consumed by the rest of my life to deal with my health issues. And pharma has replied accordingly by doing what they do best with all their R&D dollars.
I think both sides need to absorb part of the blame for the overmedication of our culture, and blaming it on modern medicine, pharma, and fancy advertising basically absolves health-care consumers of the ever-present need to be educated on matters and options concerning their health. Telling people that they remain duped by big pharma in our era of socialized education and the internet age is kind of like telling people that they are persistently stupid, which reinforces the idea that healthcare consumers may be too stupid to make their own decisions concerning matters of their health… which makes them, in turn, too trusting of anyone in the room wearing a white coat or scrubs. I trust that most adults can figure it out if they really wanted to, and could make their choices accordingly, and the last thing health care consumers need is yet another healthcare group telling them… whoops, advising them… “what to do” with their health.
I fancy living in an ideal world where all people respect and understand prevention. I don’t think disease or health issues would be eradicated, but I think a lot more of us would be healthier in general, and have fewer comorbidities when we are struck with a serious health problem. Sadly, across the board, not many are interested in this concept (government, corporations, even consumers), except for those who care to take the time to understand “alternative” medicine & the limits of modern medicine.
As someone who has lived with chronic health issues that have been remidiated by meds, I have to admit, I’m pretty frustrated with the all or nothing attitude out there about approaches to healthcare… you could say I’m “chronically” frustrated.
The reality is that some things are delt with *easiest* through medications. Yes, it’s very true that lifestyle adjustments could solve the problem, such as proper nutrition, the ability to exercise, stress resolution techniques, etc.; and yet “lifestyle adjustments” are often costly and require a certain amount of time, motivation, and education. For some people, the costs of a healthy lifestyle, believe it or not, are prohibitive in our modern culture (in a world where time, even, is money), and could be considered a luxury of the bourgoisie. How many of us are even solidly middle class today… in our current economic climate? And we know our governments are not interested in footing the bill for such fripperies as “good health” and that conceptual notion of “wellbeing.” I just wish this side of the coin would be acknowledged (the cost of preventative/alternative health) before we even step in to criticize 1) big pharma & medicine in general and 2) the individuals who choose the path of least resistance. (Yes, some people do take the short cut so they can just get on with their day, and yes, sometimes these people are looked down upon by alterna-nutz who can be equally self-righteous as the most devoted of religious zealots.)
Some conditions require meds of some sort, and for others, there are equally useful “alternative” therapies. And the reality is many conditions would simply never happen if we would only take on a preventative lifestyle and lived in a world that was a little more understanding of basic human needs & happiness! The truth is, we don’t live in an ideal world. We have the world we have today; which is a world of plurality & mass confusion what with our new toys… erm… technology. Live and let live, across the board, and let people choose the healthcare therapies that work best for them. We must all be skeptics, since that makes the healthiest of citizens, but promoting paranoia just makes people bitter at one another.
Anyways, I think my point is, the all or nothing attitude is as big a frustration coming from the realm of alternative care as it is when it comes from the medical model. Both sides need to find middle ground or they will forever be divided and we’ll waste far too much energy trying to invalidate one another, when really, there are ways for these therapies to work together. (Alternative therapies, for example, are based heavily on prevention and lifestyle, something which the medical model is only beginning to absorb.)
I think that medical science is still in its pre-historic time. And even medical technology. For example, just think how medical science will deal with these <a href=”https://www.biology-questions-and-answers.com/disease.html”>diseases</a> in the future (not near future, but in 200 to 300 year).
Thanks for your comment, Bryan. The pharmaceutical companies have played an active role in turning people into “medical consumers”. Their strategy has three primary tactics:
These efforts have been collectively recognized as a phenomenon called “Disease Mongering”. Disease mongering is the selling os sickness that widens the boundaries of illness and grows the markets for those who sell and deliver treatments.
I’ll be writing an article about disease mongering very soon!
Chris
Chris, you hit the nail on the head with this one. Our nation is over-dosing on drug abuse. People want that magic pill to make them well and drug companies are happy to oblige and do everything they can to perpetuate the myth, in the name of the almighty dollar. We need to teach people that food is our best “medicine”. Proper diet will correct most chronic diseases. I’m 56 and my goal is to have a long, healthy, and drug-free life, except for a few beers on the weekend 🙂